Answer:
Option B
Explanation:
Option (b) shows optical isomerism
$\left[Co(en)_{3}\right]^{3+}$
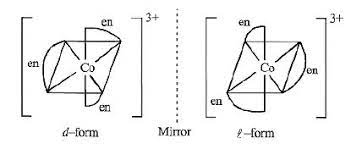
Complexes of Zn++ cannot show optical isomerism as they are tetrahedral complexes with plane of symmetry
$\left[Co(H_{2}O)_{4}(en)\right]^{3+}$have two planes of symmetry hence it is also optically inactive.
$\left[Zn(en)_{2}\right]^{2+}$cannot show optical isomerism