Answer:
Option A,B,C
Explanation:
Note: This question can be solved if the right-hand side chamber is assumed to open so that its pressure remains constant even if the piston shifts towards night.
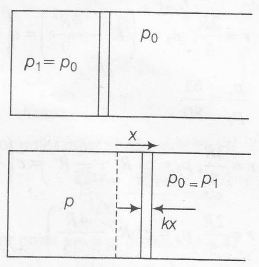
(a) pV=nRT⇒p∝TV
Temperature is made three times and volume is doubled
⇒ p2=32p1
Further x=△VA=V2−V1A
=2V1−V1A=V1A
p2=3p12=p1+kxA
⇒ KX=P1A2
Energy of spring
12kx2=p1A4x=p1V14
(b) △U=ncv△T=n(32R)△T
= 32(p2V2−p1V1)
32[(32P1)(2V1)−p1V1]=3p1V1
(c) p2=4p13⇒p2=43p1=p1+kxA
⇒ kx=p1A3⇒x=△VA=2V1A
Wgas=(p0△V+Wspring)
=(p1Ax+12kx.x)
=+(p1A.2V1A+12.p1A3.2V1A)
=2p1V1+p1V13=7p1V13
(d)
△Q=W+△U
= 7p1V13+32(p2V2−p1V1)
= 7p1V13+32(43p1.3V1−p1V1)
= 7p1V13+92p1V1=41p1V16
Note: △U=32(p2V2−p1V1) , has been obtained in part (b).