Answer:
Option C
Explanation:
In CsCl,Cl- lie at corners of simple cube and Cs+ at the body centre . Hence , along the body diagonal , Cs+ and Cl- touch each other so,
rCs++rCl−=2r
Calculation of τ
In △EDF
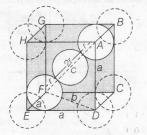
Body centred cubic unit cell
FD=b=√a2+a2=√2a
In △AFD,
c2=a2+b2
=a2+(√2a)2
=a2+2a2
c2=3a2
c=√3a
As △AFD is an equilateral triangle,
∴ √3a=4r
⇒r=√3a4
Hence, rCs++rCl−=2r
= 2×√34a=√32a